FDA Approves New Imaging Drug to Help Identify Ovarian Cancer Lesions
The U.S. Food and Drug Administration (FDA) has granted approval for Cytalux® (pafolacianine) for use in adults with ovarian cancer.1,2
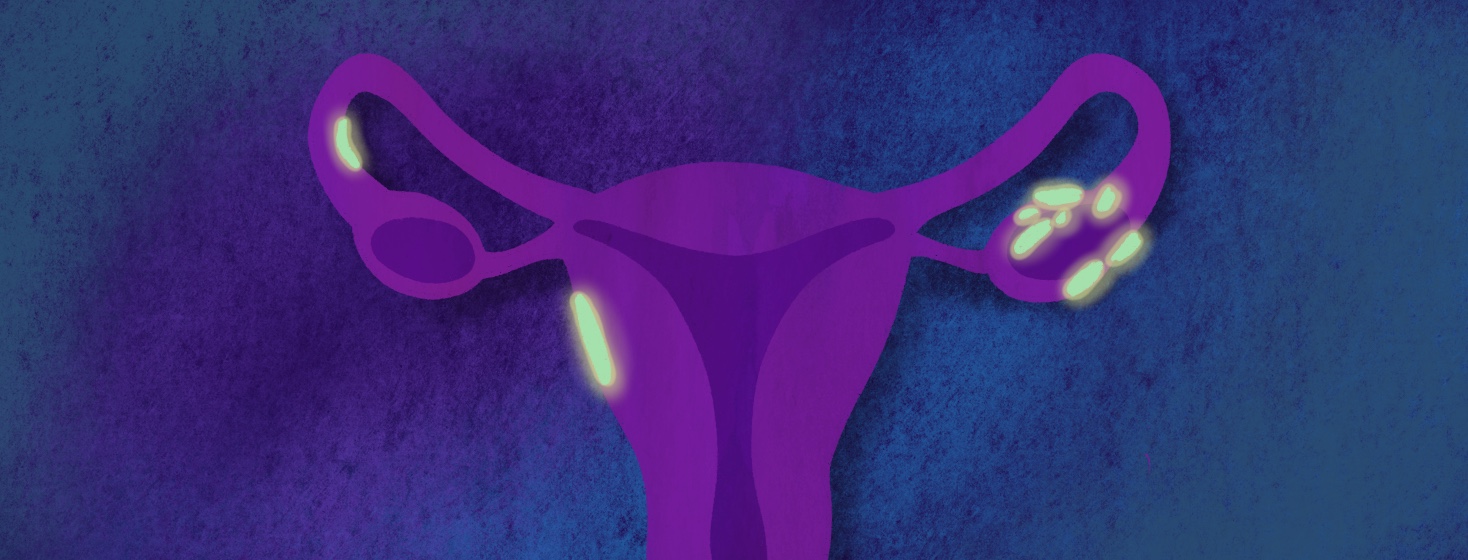
Cytalux is an imaging drug developed to help surgeons better locate cancerous lesions in cases of ovarian cancer. Cytalux illuminates under fluorescent light when it is in contact with cancerous tissue.1,2
Under current surgical protocols, doctors rely on presurgical imaging and visual or physical exams of tumors using normal light. Cytalux is an important development for people with ovarian cancer because it makes it easier for surgeons to identify lesions and reduces the risk of missing small or hidden tumors.1,2
There are often no early warning signs of ovarian cancer, and there are no standard screening tests. This means most cases of ovarian cancer are usually diagnosed in advanced stages. Treatment usually requires surgery followed by chemotherapy. However, ovarian cancer lesions can be hard to detect during surgery, especially amid organ damage and scar tissue.1,2
What are the ingredients in Cytalux?
The active ingredient in Cytalux is the optical imaging agent pafolacianine sodium.3
How does Cytalux work?
Cytalux targets a specific protein called a folate receptor found in cell membranes. Ovarian cancer causes the overproduction of this protein.1
Following injection into the body, Cytalux binds to folate receptor proteins, causing them to illuminate under fluorescent light. This allows surgeons to more accurately identify cancerous lesions.1
Evidence for Cytalux
Cytalux was studied in a randomized, multi-center phase 3 clinical trial. The study included 134 women, ages 33 to 81, who had been diagnosed with or were highly suspected to have ovarian cancer.1,2
All the women received Cytalux before surgery. They were then evaluated under both normal and fluorescent light during surgery. Nearly 27 percent of the women had at least 1 cancerous lesion detected that was not discovered by standard visual or physical exams during surgery, and on tissue that was not planned for removal.1,2
What are the possible side effects of Cytalux?
The most common side effects of Cytalux include:3
- Nausea
- Vomiting
- Abdominal pain
- Flushing
- Indigestion
- Chest discomfort
- Itching
These are not all the possible side effects of Cytalux. Talk to your doctor about what to expect when taking Cytalux. You also should call your doctor if you have any changes that concern you when taking Cytalux.
Things to know about Cytalux
There is a risk of image interpretation errors with the use of Cytalux to detect ovarian cancer during surgery. This includes false negatives and false positives.1
If you take folate, folic acid, or supplements with folate, you must stop taking these vitamins or supplements 48 hours before receiving Cytalux.3
Cytalux can harm an unborn baby. If you can become pregnant, you will be given a pregnancy test before receiving Cytalux to verify whether you are pregnant.3
Before beginning treatment for ovarian cancer, tell your doctor about all your health conditions and any other drugs, vitamins, or supplements you are taking. This includes over-the-counter drugs.
For more information, read the full prescribing information of Cytalux.
Join the conversation